 Safety
Safety
Optimized for pharmacovigilance
Designed by pharmacovigilance experts to improve operational efficiency.
Request a Demo >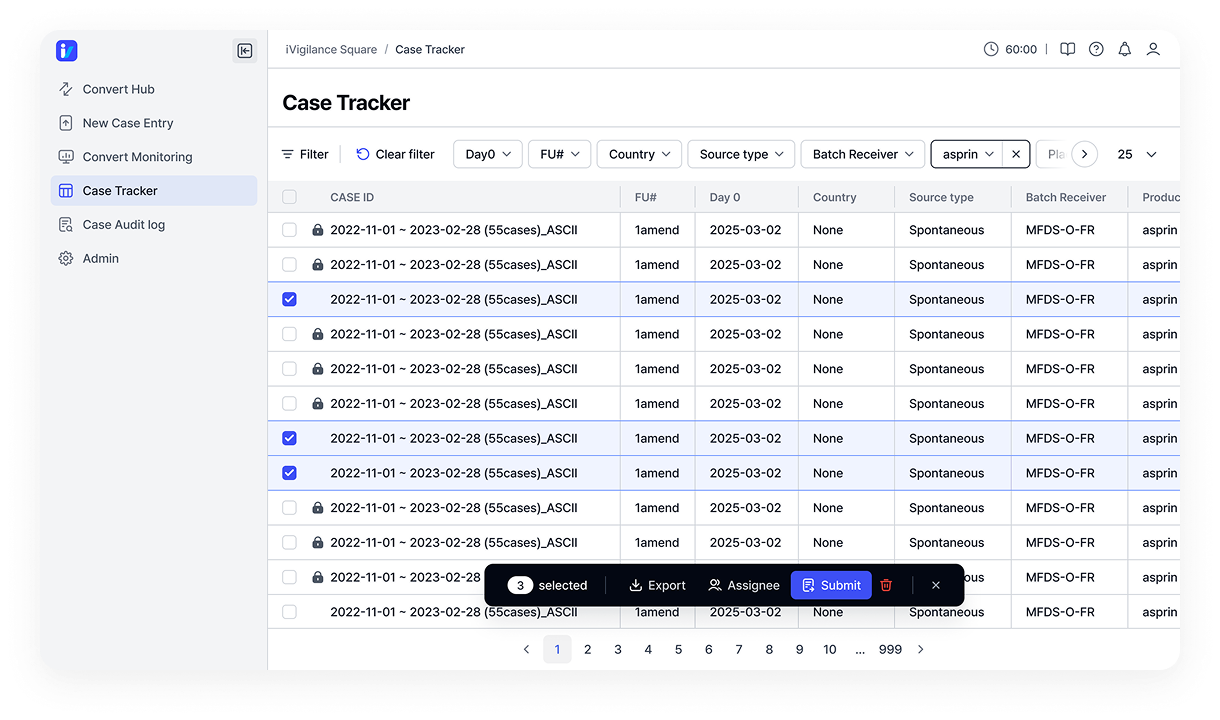

Clear visibility to support case review and decisions.

Intuitive monitoring by AE type and product

Real-time tracking of reporting status

Clear visibility to support case review and decisions.

CIOMS Report · SAE Report

Foreign R3 XML · KAERS DB

Clear mapping between AI fields and source data.

At-a-glance visibility of key fields across source and DB

Search, copy, and directly update database fields from the source
