 iVigilance Square
iVigilance Square
* PVaaS(Pharmacovigilance as a Service)
AI/DX-powered PVaaS platform delivering integrated capabilities across the entire Safety data flow—from monitoring and standardization to analytics and risk management.
Request a Demo >
Data Entry & Reporting
Delivers AI-driven data entry for CIOMS and SAE reports, and provides robust MFDS-compliant submission capabilities for all foreign adverse event cases.

Automated Literature Monitoring
Executes automated literature searches and logging with a single query, while AI summarizes full texts and automatically detects ICSR candidates from narrative content.

For MFDS Product Renewal Report
Provides guideline-compliant tabulation for MFDS Product Renewal Reports and high-volume automated analytics for KAERS data.
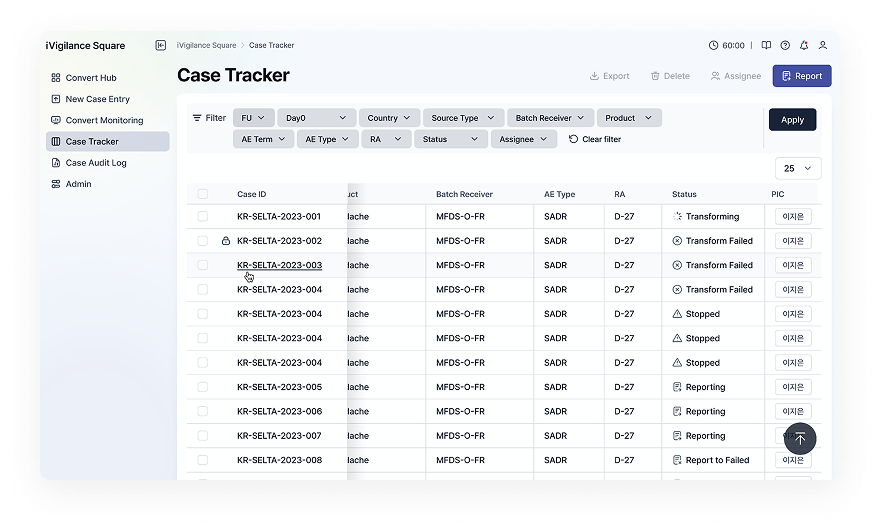
Standardizing data into a Safety Data structure, and ICSR processing/reporting
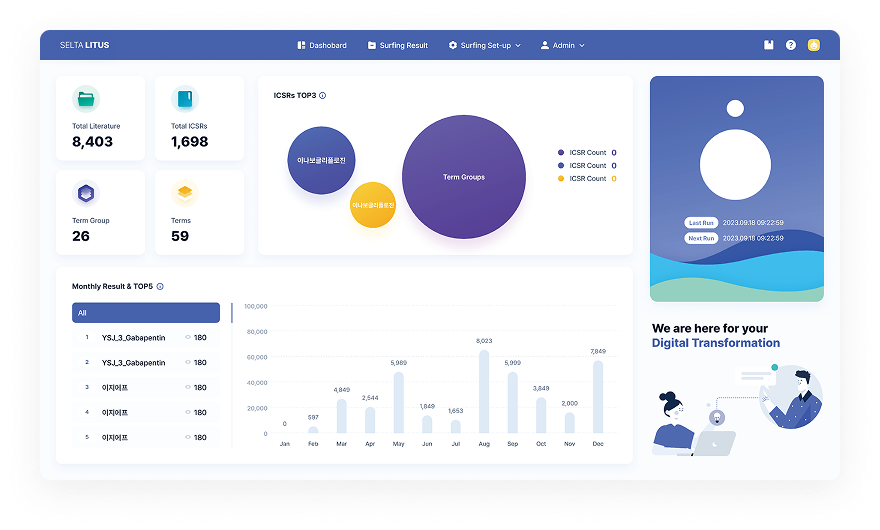
Literature Monitoring Automation & AI-driven Safety Valid Case Detection
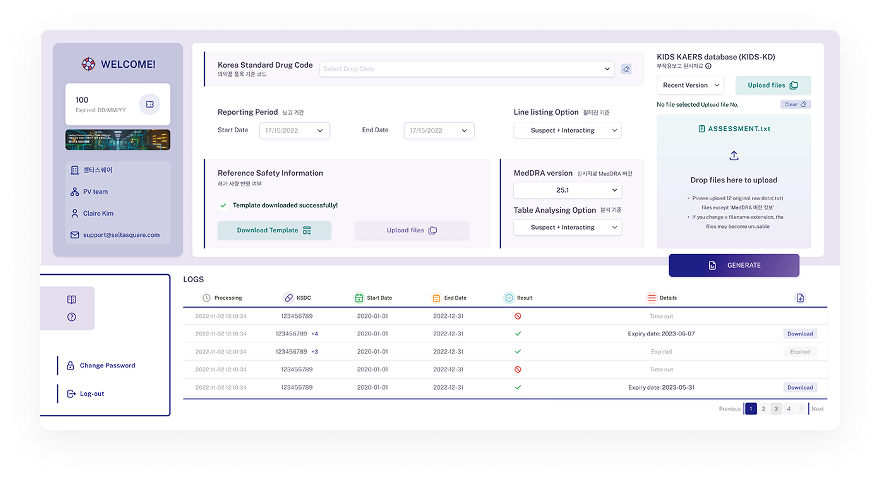
Instant analysis results provided with a single click, even for large volumes of data
